 Breeding season is just around the corner and most breeders dread the frustration of subfertility in certain broodmares. A thorough breeding history is key to diagnosing the cause of subfertility. Questions that address previous breeding management, stallion selection, and general wellness are important when performing a breeding soundness evaluation. Diagnostics such as trans-rectal palpation and ultrasound examination of the reproductive tract, vaginal speculum examination, digital cervical palpation, endometrial cytology and culture, endometrial biopsy, and hysteroscopic examination are useful in determining the cause of subfertility. Typically, a cause of subfertility can be determined utilizing the diagnostics mentioned above, but there may be some cases in which diagnostic test results are negative and yet a mare is still subfertile. So what’s next?
Breeding season is just around the corner and most breeders dread the frustration of subfertility in certain broodmares. A thorough breeding history is key to diagnosing the cause of subfertility. Questions that address previous breeding management, stallion selection, and general wellness are important when performing a breeding soundness evaluation. Diagnostics such as trans-rectal palpation and ultrasound examination of the reproductive tract, vaginal speculum examination, digital cervical palpation, endometrial cytology and culture, endometrial biopsy, and hysteroscopic examination are useful in determining the cause of subfertility. Typically, a cause of subfertility can be determined utilizing the diagnostics mentioned above, but there may be some cases in which diagnostic test results are negative and yet a mare is still subfertile. So what’s next?
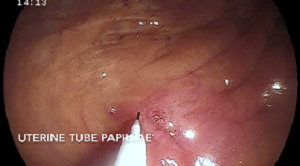
Although rare, uterine tube (oviduct) abnormalities have been described in mares and can cause subfertility. The uterine tube consists of the infundibulum (fan-like structures that accepts the ovum at the time of ovulation), ampulla (site of fertilization), isthmus, and uterine tube papilla (opens into the uterine lumen). Uterine tube abnormalities that have been described include adhesions, inflammation/infection (salpingitis), neoplasia, cysts, fluid accumulation (hydrosalpinx), and uterine tube obstructions. Typically, these mares present with subfertility or infertility without a known cause. Clinical diagnosis of these abnormalities is difficult; therefore, a diagnosis of exclusion is usually made. In research settings, uterine tube patency can be tested with the placement of microspheres into the uterine tubes or starch granules onto the surface of the ovary and subsequent confirmation of their presence within the uterus[1,2]. 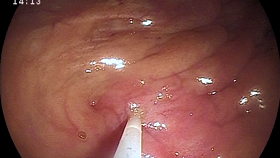 These methods are not easily performed in the clinic; therefore, presumptive diagnoses of uterine tube abnormalities have been made when all other potential causes of subfertility have been ruled out, making this often a diagnosis of exclusion in most cases of subfertility. Several studies performed on reproductive tracts obtained from slaughterhouses have described gelatinous masses within the uterine tube of mares of all ages[3-6]. These masses are suspected to cause a partial or complete obstruction of the lumen of the uterine tube and interfere with fertilization, sperm, ovum, or embryo transport[3-6]. Several treatment options for uterine tube obstructions have been utilized over the years to resolve this presumed condition. Techniques developed to treat these pathologies include retrograde and normograde catheterization of the uterine tube via laparotomy, laparoscopic application of prostaglandin E2 (PGE2) to the uterine tube, hysteroscopic hydrotubation of the uterine tube, and application of misoprostol (prostaglandin E1) to the uterine tube papilla[7-11].
These methods are not easily performed in the clinic; therefore, presumptive diagnoses of uterine tube abnormalities have been made when all other potential causes of subfertility have been ruled out, making this often a diagnosis of exclusion in most cases of subfertility. Several studies performed on reproductive tracts obtained from slaughterhouses have described gelatinous masses within the uterine tube of mares of all ages[3-6]. These masses are suspected to cause a partial or complete obstruction of the lumen of the uterine tube and interfere with fertilization, sperm, ovum, or embryo transport[3-6]. Several treatment options for uterine tube obstructions have been utilized over the years to resolve this presumed condition. Techniques developed to treat these pathologies include retrograde and normograde catheterization of the uterine tube via laparotomy, laparoscopic application of prostaglandin E2 (PGE2) to the uterine tube, hysteroscopic hydrotubation of the uterine tube, and application of misoprostol (prostaglandin E1) to the uterine tube papilla[7-11].
Zent et al. (1993) catheterized and flushed the uterine tube in a retrograde manner via laparotomy successfully in a group of barren mares. Bennet et al. (2002) described normograde catheterization and flushing of the uterine tube via laparotomy to determine uterine tube patency and treat uterine tube occlusion. Eight of 12 mares, that had been barren for 2-4 years, became pregnant following the procedure[8]. Prostaglandin E 2 is produced by the equine embryo starting at day 5 post ovulation[12-14]. This hormone binds to receptors on smooth muscle cells in the uterine tube and allows for embryo transport within the uterine tube[12-14]. Laparoscopic application of PGE2 to the uterine tube has been utilized successfully to treat subfertile mares in which a cause of subfertility was not identified[7,15]. This technique also allows for visualization of the uterine tube and identification of pathologies such as distension, masses, or adhesions[1,7,15].
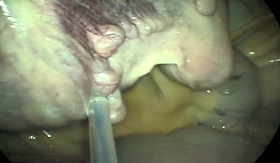 Uterine tube hydrotubation is a procedure in which the uterine tube papilla is catheterized and flushed with sterile isotonic solution in a retrograde direction via hysteroscopy[9,16]. This procedure was developed by Dr. Yuji Inoue and has gained popularity in recent years as a method to successfully treat subfertile mares in which a cause for subfertility has not been identified[9]. Inoue et al. (2018) reported that 92.8% (26 out of 28) of subfertile mares that had this procedure performed became pregnant after this treatment. This technique allows for visualization of the vagina, cervix, and uterine lumen. This may offer a diagnostic advantage over the laparoscopic procedure as uterine pathologies, not typically diagnosed with typical palpation and ultrasound of the reproductive tract, may be visualized with this technique. Alvarenga et al. (2018) described a new technique in which misoprostol (synthetic prostaglandin E1) is applied to the uterine tube papilla utilizing deep horn insemination technique. Mares had been barren for at least 2 years and 72.2% (16/22) of mares became pregnant by at least 3 cycles after the procedure. This technique is easily performed and is less expensive than that of the other procedures mentioned above. Additionally, this procedure can be performed in the field by anyone comfortable with deep-horn insemination technique, making it a treatment available to a larger group of practitioners.
Uterine tube hydrotubation is a procedure in which the uterine tube papilla is catheterized and flushed with sterile isotonic solution in a retrograde direction via hysteroscopy[9,16]. This procedure was developed by Dr. Yuji Inoue and has gained popularity in recent years as a method to successfully treat subfertile mares in which a cause for subfertility has not been identified[9]. Inoue et al. (2018) reported that 92.8% (26 out of 28) of subfertile mares that had this procedure performed became pregnant after this treatment. This technique allows for visualization of the vagina, cervix, and uterine lumen. This may offer a diagnostic advantage over the laparoscopic procedure as uterine pathologies, not typically diagnosed with typical palpation and ultrasound of the reproductive tract, may be visualized with this technique. Alvarenga et al. (2018) described a new technique in which misoprostol (synthetic prostaglandin E1) is applied to the uterine tube papilla utilizing deep horn insemination technique. Mares had been barren for at least 2 years and 72.2% (16/22) of mares became pregnant by at least 3 cycles after the procedure. This technique is easily performed and is less expensive than that of the other procedures mentioned above. Additionally, this procedure can be performed in the field by anyone comfortable with deep-horn insemination technique, making it a treatment available to a larger group of practitioners.
There are several options for the treatment of presumed uterine tube abnormalities that have increased pregnancy rates in previously barren mares. A retrospective study comparing pregnancy rates after application of PGE2 to the uterine tube versus pregnancy rates after hysteroscopic hydrotubation of the uterine tubes was performed in 2018 (Walbornn et al. 2018; AAEP Proceedings). These two procedures are typically performed at our hospital to treat presumptive uterine tube obstruction; therefore, we were interested in whether one treatment could achieve better pregnancy rates than the other. 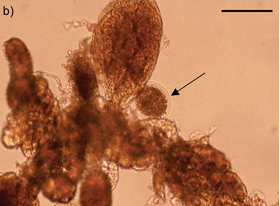 No significant difference (P>0.05) was found between pregnancy rates compared within this study. Decision on treatment can be based on comfort level of the practitioner with either procedure, proximity to a referral clinic, cost, and a mare’s purpose (broodmare vs. sport horse).
No significant difference (P>0.05) was found between pregnancy rates compared within this study. Decision on treatment can be based on comfort level of the practitioner with either procedure, proximity to a referral clinic, cost, and a mare’s purpose (broodmare vs. sport horse).
Unexplained subfertility can be frustrating and challenging for owners and veterinarians alike. A thorough breeding soundness evaluation should be the first step to diagnosing the cause of subfertility in broodmares. If no cause can be identified, the adjunctive treatment options outlined above can be utilized to treat presumptive uterine tube abnormalities and potentially increase pregnancy rates in previously subfertile mares.
_________________________________________________________________________________________
Image 1 (top left): Catheter on the left side of the uterine tube papilla with guide wire emerging from the tip, seen via hysteroscopy.
Image 2 (middle right): Catheter placed within uterine tube papilla and guide wire removed, seen via hysteroscopy.
Image 3 (middle left): View of ovary and uterine tube via laparoscopy and pipette placing PGF2a on the serosal surface of uterine tube. (*Image courtesy of Dr. Patrick McCue)
Image 4 (bottom right): Uterine cells and oocyte recovered from uterine flushing post PGF 2application to uterine tube. Black arrow is pointing to oocyte. (Allen et al. 2016)
__________________________________________________________________________________________
[1] Arnold CE, Love CC. Laparoscopic evaluation of oviductal patency in the standing mare. Theriogenology. 2013;79:905-10.
[2] Dascanio JJ, McCue PM. Equine Reproductive Procedures. In: John Wiley & Sons I, editor.2014.
[3] Onuma H, Ohnami Y. Retention of tubal eggs in mares. J Reprod Fertil Suppl. 1975:507-11.
[4] Tsutsumi Y, Suzuki H, Takeda T, Terami Y. Evidence of the origin of the gelatinous masses in the oviducts of mares. J Reprod Fertil. 1979;57:287-90.
[5] van Niekirk CH, Gerneke WH. Persistence and Parthogenetic Cleavage of Tubal Ova in the Mare. Onderstepoort J Vet Res. 1966;33:195-232.
[6] Aguilar JJ, Woods GL, Miragaya MH, Olsen LM. Living fibroblast cells in the oviductal masses of mares. Equine Vet J Suppl. 1997:103-8.
[7] Allen WR, Wilsher S, Morris L, Crowhurst JS, Hillyer MH, Neal HN. Laparoscopic application of PGE2 to re-establish oviducal patency and fertility in infertile mares: a preliminary study. Equine Vet J. 2006;38:454-9.
[8] Bennet S, Griffin R, Rhoads W. Surgical Evaluation of the Oviduct Disease and Patency in the Mare. AAEP Proceedings 2002. 2002;48:347-9.
[9] Inoue Y, Sekiguchi M. Clinical application of hysteroscopic hydrotubation for unexplained infertility in the mare. Equine Vet J. 2017.
[10] Alvarenga MA, Segabinazzi LG. Application of Misoprostol as a Treatment of Unexplained Infertility in Mares. Journal of Equine Veterinary Science. 2018;71:46-50.
[11] Zent WW, Liu KM, Spirito MA. Oviduct flushing as a treatment for infertility in the mare. Equine Vet J. 1993;15:47-8.
[12] Weber JA, Freeman DA, Vanderwall DK, Woods GL. Prostaglandin E2 secretion by oviductal transport-stage equine embryos. Biol Reprod. 1991;45:540-3.
[13] Weber JA, Freeman DA, Vanderwall DK, Woods GL. Prostaglandin E2 hastens oviductal transport of equine embryos. Biol Reprod. 1991;45:544-6.
[14] Weber JA, Woods GL, Freeman DA, Vanderwall DK. Prostaglandin E2 secretion by day-6 to day-9 equine embryos. Prostaglandins. 1992;43:55-9.
[15] Ortis HA, Foss RR, McCue PM, Bradecamp EA, Ferris RA, Hendrickson DA. Laparoscpic Application of PGE2 to the Uterine Tube Surface Enhances Fertility in Selected Subfertile Mares. Journal of Equine Veterinary Science. 2013;33:896-900.
[16] Schnobrich M. How to Use Endoscopy (Hysteroscopy and Laparoscopy) to Manage the Sub-Fertile Mare. American Association of Equine Practitioners. Orlando, FL2016. p. 169-74.


Log in to join the conversation.